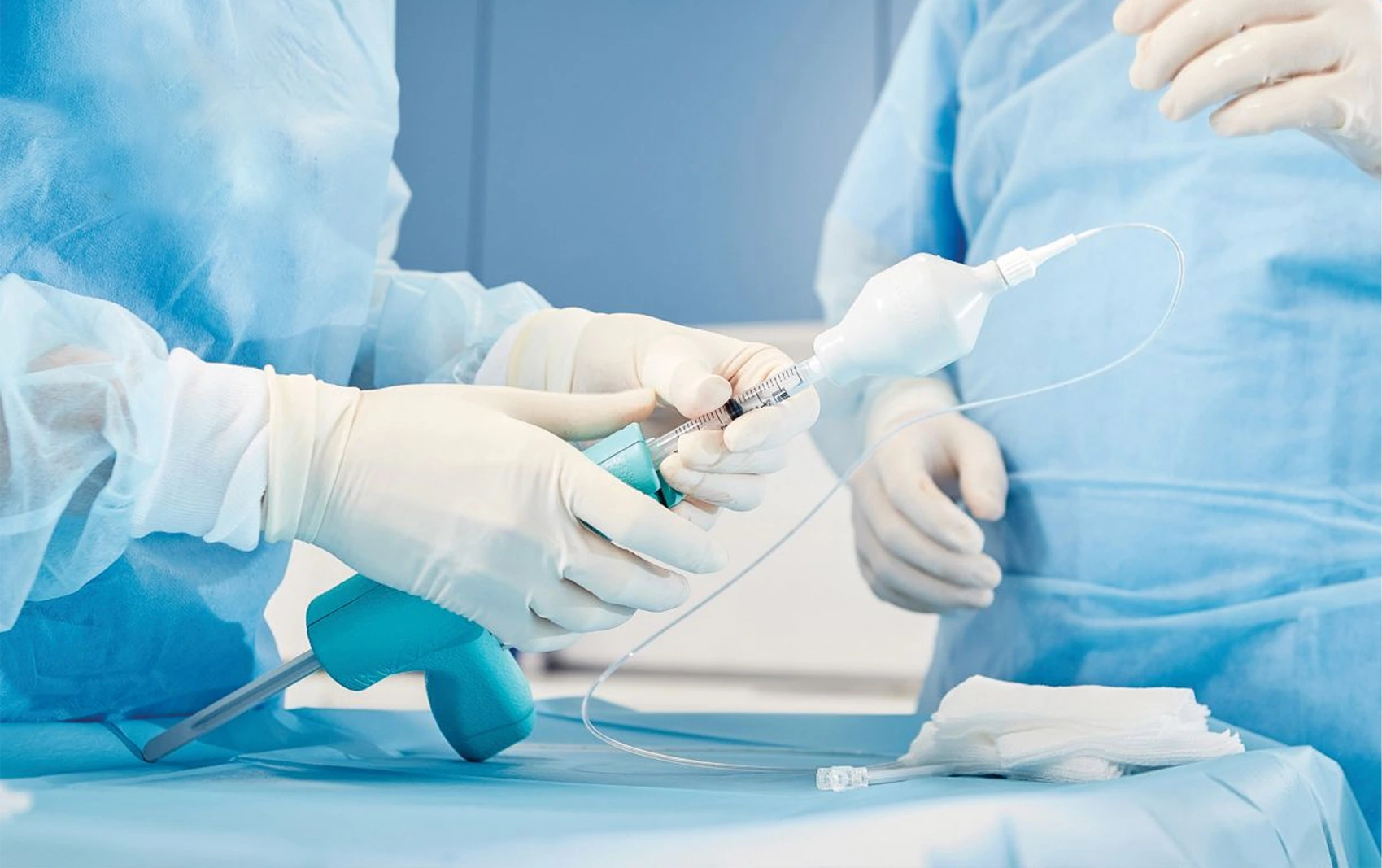
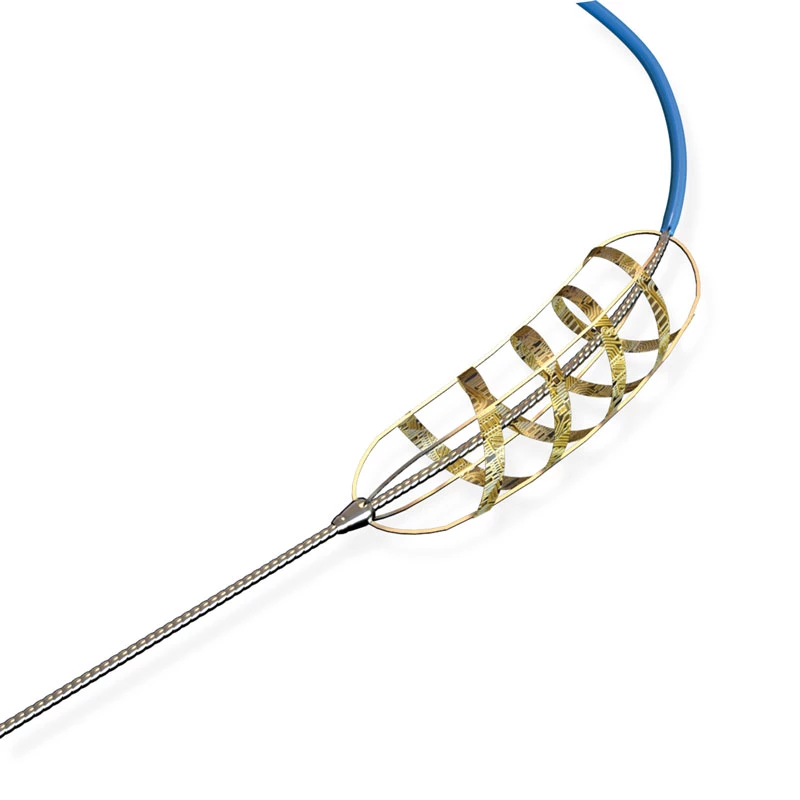
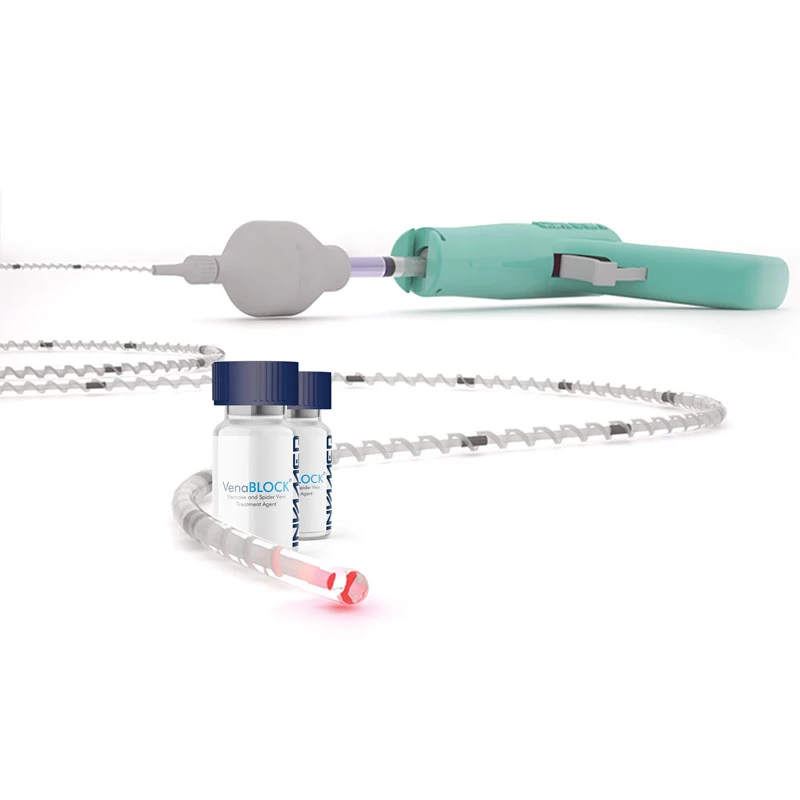
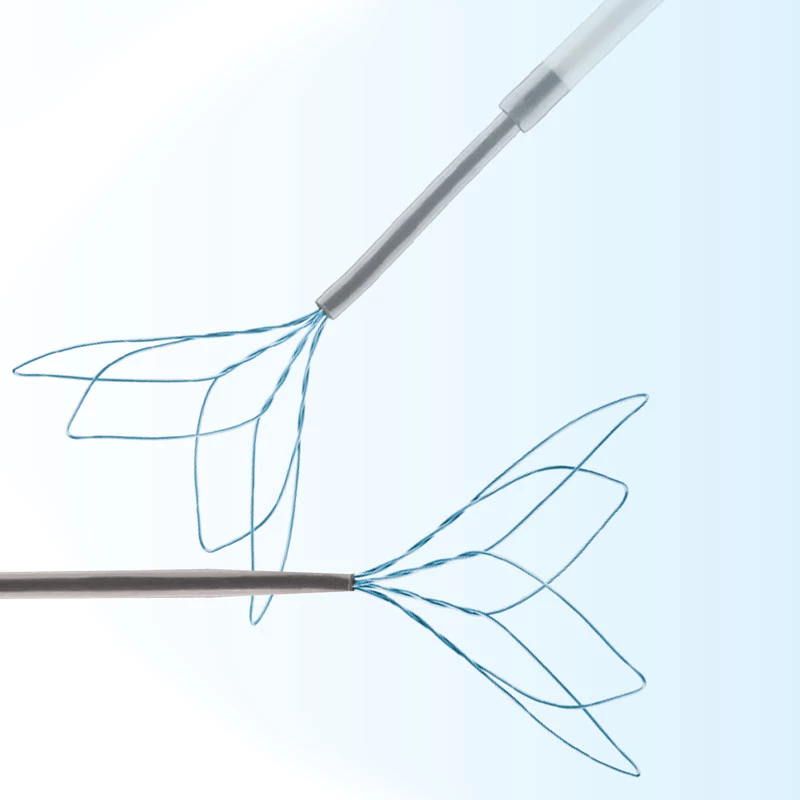
Who we are?
Setting out to meet the needs in the healthcare field with high-end engineering products, INVAMED adopts the slogan/motto "Innovations Are Born At Invamed" and offers patient-oriented solutions with over hundred internationally patented medical devices of high quality and world standards. Strong positive feedback from our clinical trials are equally reflected in our strong innovation goals. We use all our strength to advance our groundbreaking treatment solutions in healthcare technology.
With our unique treatment techniques, we have achieved many successes that go beyond the borders of the country. Thanks to our treatment methods, our products are the first choice for doctors as they provide longer-term results.
By increasing the operability of existing resources and manufacturing facilities, we have maximized our technical possibilities for the projects of our entrepreneurial R&D team to turn into products.


Our Products
We have dedicated to continuous innovation in order to shape everything we do.